Abstract
Spontaneous regression of chronic lymphocytic leukemia occurs in less than 2% of cases and the mechanisms that underlie this rare event remain largely unknown. We reviewed 1425 patients with CLL across 4 centers and identified 20 who underwent spontaneous disease regression without treatment over a median period of 8.6 years. This included 12 patients with complete and 8 with partial (> 50%) resolution of lymphocytosis and clinical features. Residual circulating CLL cells remained detectable in all cases and phenotypic, functional and genomic analyses were performed on the residual CLL clone, as well as stored cells where available, to investigate potential mechanisms underlying regression.
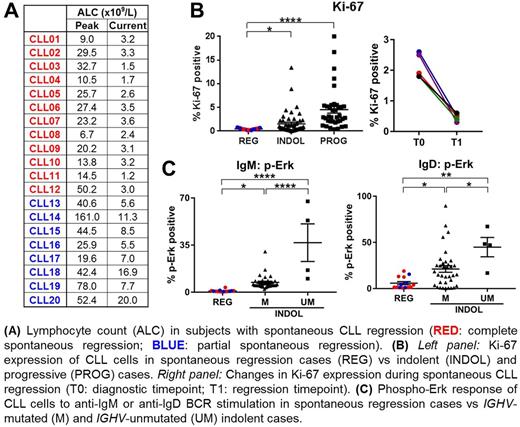
IGH sequence analysis of sequential samples confirmed that the CLL clone was identical between the time of diagnosis and regression (n=6). Moreover, all cases showed somatic mutation of the IGHV gene and VH3 or VH4 usage was seen in 17 cases. Flow cytometric analysis of spontaneous regression cases (n=17) showed that Ki-67 and CD49d expression was markedly decreased compared to stable indolent disease (n=54) and progressive disease (n=40) whereas CXCR4 and FasR expression was increased. No difference was observed with respect to the expression of CD38, ZAP-70, Bcl-2 and Mcl-1 between spontaneous regression and indolent cases.
Sequential analysis revealed that these changes in Ki-67 and CXCR4 expression developed during the period of regression. Furthermore, telomere length analysis demonstrated that tumor cells generally had short telomeres (< 5 kb) after regression and this profile, together with the phenotypic progression, indicates that tumors have undergone an initial period of intense proliferation prior to the development of regression. Importantly, telomerase expression remained high in regression tumors and no evidence of cellular senescence was observed. Analysis of the immune compartment revealed a reduction in T cell and NK cell numbers during spontaneous CLL regression and a modest improvement in T cell function as revealed by increased T cell proliferation and decreased expression of PD-1.
The response of tumor cells to stimulation through the B cell receptor (BCR) was also investigated. Surface expression of IgM and IgD was low in regression cases and basal Erk phosphorylation was increased. In addition, these tumors were uniformly unresponsive to stimulation separately with anti-IgM F(ab')2 antibodies and anti-IgD F(ab')2 antibodies, and combined stimulation with both antibodies. These findings indicate that spontaneously regressed CLL tumors are in a state of profound unresponsiveness to BCR stimulation. Consistent with reduced BCR responsiveness, RNA-seq analysis of sorted CLL cells revealed reduced expression of both MYC and Myc target genes in regression cases as well as downregulation of pathways involved in RNA processing, cellular metabolism and proliferation.
Finally, the genomic landscape of tumor cells after regression was analyzed by whole exome sequencing and SNP array analysis on paired germline and CLL samples. CLL genomic complexity was generally low although mutations in TP53, MYD88 or HIST1H1B were observed in 5 tumors. Strikingly, 13q14 duplication was present in 13 of 19 tumors and qPCR analysis showed that this was associated with increased expression of miR-15a/16-1.
This is the largest and most comprehensive analysis of spontaneous CLL regression to date and reveals a range of novel features. Tumors that have undergone spontaneous regression express somatically mutated immunoglobulin genes, demonstrate short telomeres with a phenotype of BCR unresponsiveness and have a very high prevalence of 13q14 duplication. This profile suggests a model in which CLL clones undergo substantial proliferation initially which subsequently subside into a state of BCR unresponsiveness and low proliferation. The identification of 13q14 duplication as a recurrent genetic abnormality indicates a potential molecular mechanism for this pattern but it is currently unclear if this lesion is established during tumorigenesis or is acquired during the initial proliferative phase. These findings identify CLL regression as a unique subset of B cell lymphoproliferative disease and suggest that therapies which upregulate the expression of miR-15a/16-1 may represent a mechanism to suppress B-CLL proliferation.
Rawstron: Janssen: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; BD biosciences: Patents & Royalties; Roche: Consultancy, Honoraria; Gilead: Research Funding. Munir: Alexion Pharmaceuticals, Inc.: Honoraria; Roche: Honoraria; Gilled: Honoraria; Janssen: Honoraria; AbbVie: Honoraria. Forconi: AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Infinity: Consultancy, Membership on an entity's Board of Directors or advisory committees; Gilead: Research Funding; Janssen-Cilag: Speakers Bureau. Hillmen: AbbVie: Consultancy, Honoraria, Research Funding; Roche: Consultancy, Honoraria, Research Funding; Pharmacyclics LLC, an AbbVie Company: Honoraria, Research Funding; Gilead: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Alexion Pharmaceuticals, Inc.: Consultancy, Honoraria; Novartis: Honoraria, Research Funding; Celgene: Research Funding; GSK: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.